Chronic myeloid leukemia (CML) and its peer markets - Hodgkin lymphoma, Acute myeloid leukemia (AML), Non-Hodgkin lymphoma, Acute lymphoblastic leukemia (ALL), B-cell Lymphoma, Chronic lymphocytic leukemia (CLL), Multiple myeloma, Burkitt lymphoma, Adult T-cell leukemia and 3 other type markets - adds up to total Haematopoietic and lymphoid tissues Cancers market.
Chronic myeloid leukemia (CML) can be segmented by Submarkets, Technologies, Products, Geographies, Companies and Phases. Submarkets of this market are Chromatography, E-Clinical Trial Solutions, Wireless Health, Clinical Trial Management Systems (CTMS), Healthcare IT outsourcing, Immunoassay, Digital Pathology, Chromatography Instruments, Next Generation Sequencing (NGS), Proteomics, Marketed Drugs (CML) and Pipeline Drugs (CML). Technologies of this market are Microfluidics and Chromatography Reagents. Products of this market are Lab-on-chips (Microfluidics), Microreactors, AMG 103, DFP-10917, Tasigna and BP-100-1.01. Geographies of this market are USA, Canada, United Kingdom, Germany, France, Italy, Spain and Japan. Companies of this market are Pfizer Inc, Novartis AG, ARIAD Pharmaceuticals Inc and BRISTOL-MYERS SQUIBB. Phases of this market are Phase II.
Chronic myeloid leukemia (CML) can be segmented by Submarkets, Technologies, Products, Geographies, Companies and Phases. Submarkets of this market are Chromatography, E-Clinical Trial Solutions, Wireless Health, Clinical Trial Management Systems (CTMS), Healthcare IT outsourcing, Immunoassay, Digital Pathology, Chromatography Instruments, Next Generation Sequencing (NGS), Proteomics, Marketed Drugs (CML) and Pipeline Drugs (CML). Technologies of this market are Microfluidics and Chromatography Reagents. Products of this market are Lab-on-chips (Microfluidics), Microreactors, AMG 103, DFP-10917, Tasigna and BP-100-1.01. Geographies of this market are USA, Canada, United Kingdom, Germany, France, Italy, Spain and Japan. Companies of this market are Pfizer Inc, Novartis AG, ARIAD Pharmaceuticals Inc and BRISTOL-MYERS SQUIBB. Phases of this market are Phase II.
Key Questions Answered
- What are market estimates and forecasts; which of Chronic myeloid leukemia (CML) markets are doing well and which are not?
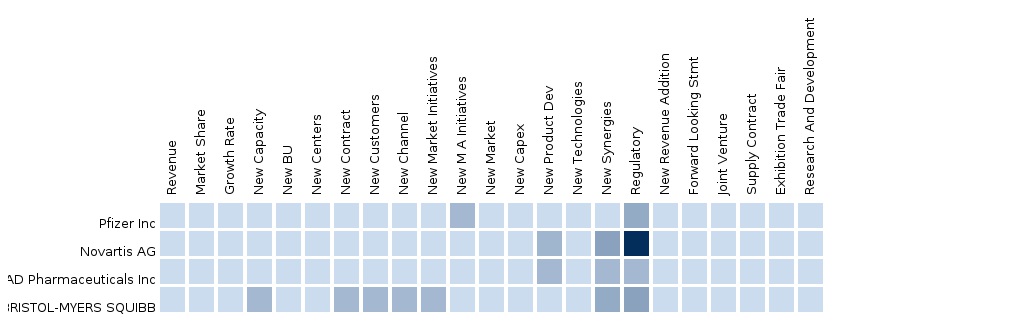
- What is the competitive landscape; How companies like Pfizer Inc, Novartis AG and ARIAD Pharmaceuticals Inc doing in Chronic myeloid leukemia (CML)?
- This reports provides most granular segmentation on Chromatography, E-Clinical Trial Solutions, Wireless Health and Clinical Trial Management Systems (CTMS).
- This report provides market sizing and forecast for the Chronic myeloid leukemia (CML) market. It also provides market sizing and forecast along with the drivers/inhibitors/opportunity analysis for each of the micro markets.
- The report provides deep dive competitive landscape covering the top players such as Pfizer Inc, Novartis AG, ARIAD Pharmaceuticals Inc and BRISTOL-MYERS SQUIBB.
- The reports provides benchmarking insight on the top players Pfizer Inc, Novartis AG, ARIAD Pharmaceuticals Inc and BRISTOL-MYERS SQUIBB.
- The report provide competitive intelligence on Pfizer Inc, Novartis AG, ARIAD Pharmaceuticals Inc and BRISTOL-MYERS SQUIBB.
- Global Chronic myeloid leukemia (CML) companies
- Manufacturing Companies
- Traders, distributors, and suppliers
- Governmental and research organizations
- Associations and industry bodies
- Technology providers
- European Medicines Agency accepted Pfizer's regulatory submissions for review of bosutinib(drug)
- Bristol-Myers and Innate Pharma S.A. (France) entered into an agreement to develop and market Phase I drug, IPH2102, human monoclonal antibody for cancer treatment
- Bristol-Myers Squibb and Otsuka Pharmaceutical Co. Ltd. declared 18-month follow-up results for Phase III DASISION study of Sprycel versus imatinib for the treatment of adults with Philadelphia chromosome-positive chronic phase chronic myeloid leukemia.
- InVivoScribe Technologies Inc. and Novartis collaborated to develop and market a companion diagnostic test
- Novartis obtained Canada healthcare authority approval for Tasigna
- Bristol-Myers Squibb and Otsuka America Pharmaceutical Inc. declared five-year follow-up results from Phase III study of Sprycel in Philadelphia
- The company collaborated with MolecularMD Corporation (U.S.)
- Novartis received approval for Tasigna as first-line therapy for the treatment of chronic myelogenous leukemia patient in Japan.
- Novartis received Japan's Ministry of Health, Labour and Welfare’s approval for Tasigna (nilotinib)
- Bristol-Myers Squibb obtained European Commission Marketing Authorization for Sprycel (dasatinib) for the treatment of adult Philadelphia chromosome positive chronic myelogenous leukemia in the chronic phase.
What makes our report unique?
Audience for this report
Top developments
1 Introduction
1.1 Analyst Insights
1.2 Market Definitions
1.3 Market Segmentation & Aspects Covered
1.4 Research Methodology
2 Executive Summary
3 Market Overview
4 By Submarkets
4.1 Chromatography
4.2 E-Clinical Trial Solutions
4.3 Wireless Health
4.4 Clinical Trial Management Systems (CTMS)
4.5 Healthcare IT outsourcing
4.6 Immunoassay
4.7 Digital Pathology
4.8 Chromatography Instruments
4.9 Next Generation Sequencing (NGS)
4.10 Proteomics
4.11 Marketed Drugs (CML)
4.12 Pipeline Drugs (CML)
5 By Technologies
5.1 Microfluidics
5.2 Chromatography Reagents
6 By Products
6.1 Lab-on-chips (Microfluidics)
6.2 Microreactors
6.3 AMG 103
6.4 DFP-10917
6.5 Tasigna
6.6 BP-100-1.01
7 By Phases
7.1 Phase II
8 By Geographies
8.1 USA
8.2 Canada
8.3 United Kingdom
8.4 Germany
8.5 France
8.6 Italy
8.7 Spain
8.8 Japan
9 By Companies
9.1 Pfizer Inc
9.2 Novartis AG
9.3 ARIAD Pharmaceuticals Inc
9.4 BRISTOL-MYERS SQUIBB
Please fill in the form below to receive a free copy of the Summary of this Report
Custom Market Research Services
We will customize the research for you, in case the report listed above does not
meet with your exact requirements. Our custom research will comprehensively cover
the business information you require to help you arrive at strategic and profitable
business decisions.
Please visit https://www.micromarketmonitor.com/custom-research-services.html to specify your custom Research Requirement
Please visit https://www.micromarketmonitor.com/custom-research-services.html to specify your custom Research Requirement
| PRODUCT TITLE | PUBLISHED | |
|---|---|---|
 |
Bosutinib Bosutinib and Lab-on-chips (Microfluidics), |
Upcoming |
 |
Gleevec Gleevec and Lab-on-chips (Microfluidics), Microreactors,... |
Upcoming |
 |
Sprycel Sprycel and Lab-on-chips (Microfluidics), Microreactors,... |
Upcoming |
 |
Phase I (CML) Phase I (CML) and Phase II, Phase II (CML) and |
Upcoming |
 |
Phase II (CML) Phase II (CML) and Phase II, Phase I (CML) and |
Upcoming |
 |
Phase III (CML) Phase III (CML) and Phase II, Phase I (CML) and |
Upcoming |
6 reports |
Show












